“ Alpha Radiation.” An Overview | ScienceDirect Topics. The key difference between helium atom and alpha particle is that helium atom has two protons, two neutrons, and two electrons, whereas alpha particle only has two protons and two neutrons. Alpha particle contains two protons and two neutrons that are bound together into a particle. Helium atom or helium chemical element is the atom having the atomic number 2 and chemical symbol He. The below infographic presents the differences between helium atom and alpha particle in tabular form for side-by-side comparison. In addition, helium atoms are more stable than alpha particles. Moreover, the helium atom is unreactive, whereas the alpha particle is reactive. However, the nucleus of the helium atom is identical to the alpha particle. Helium atom and alpha particle are different from each other according to the presence or absence of electrons. 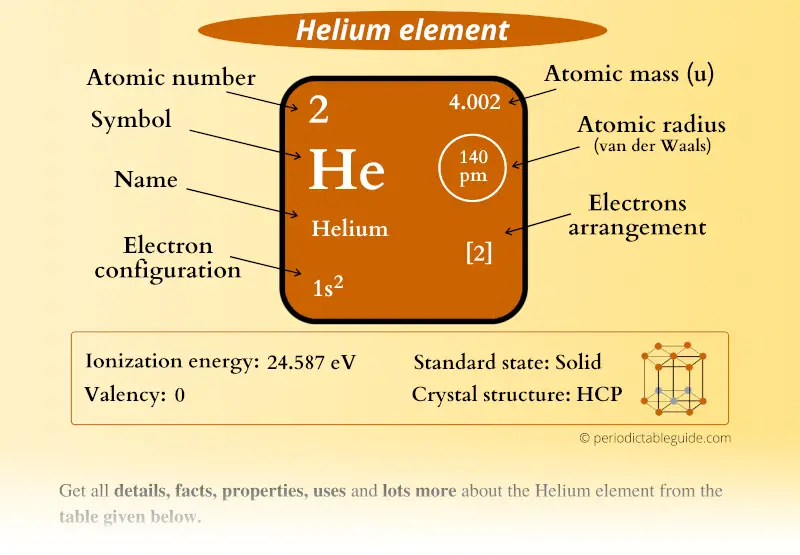
What is the Difference Between Helium Atom and Alpha Particle? Therefore, alpha particles are a highly ionizing form of particle radiation that usually have a low penetration depth. The mechanism of production of alpha particle in radioactive decay make them have a kinetic energy of around 5 MeV, and the velocity is about 4% of that of the speed of light. The net spin of the alpha particle is zero. This indicates a helium atom with a +2 charge. However, since it is identical to the nucleus of the helium atom, we can denote it as He 2+ as well. The symbol for this particle can be given as α. 
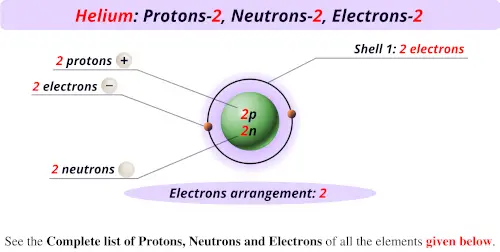
The alpha particle was named after the first letter in the Greek alphabet. Generally, these particles are formed in the process of alpha decay. This particle is identical to the helium-4 nucleus. What is Alpha Particle?Īlpha particle contains two protons and two neutrons that are bound together into a particle. This helium atomic nucleus is identical to an alpha particle. It contains two electrons in atomic orbitals that surround a nucleus consisting of two protons and two neutrons. Helium is the second simplest atom to model in quantum mechanics. This discovery was made by Georges Rayet, Captain C.T. Helium atoms were detected for the first time as an unknown, yellow spectral line signature in sunlight during a solar eclipse in 1868. At standard temperature and pressure, helium occurs as a gas. This chemical element can be found in period 1, group 18, in the periodic table of elements. It is an s block element having the electron configuration 1s2. Moreover, it is the second most abundant chemical element in the universe. The melting and boiling temperatures of helium atoms are minimal among all elements. It is a colorless, odorless, tasteless, nontoxic, inert monoatomic gas that comes as the first in the noble gas group. Summary – Helium Atom vs Alpha Particle What is Helium Atom? Helium Atom vs Alpha Particle in Tabular Formĥ. When considering the nucleus of the helium atom, it is identical to the alpha particle according to the number of protons and neutrons. The helium atom is a common chemical species in the universe.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
